
Coronavirus SARS-CoV-2 (COVID-19) Research Solutions
Accelerate your COVID-19 research
Supporting the global research community working on COVID-19

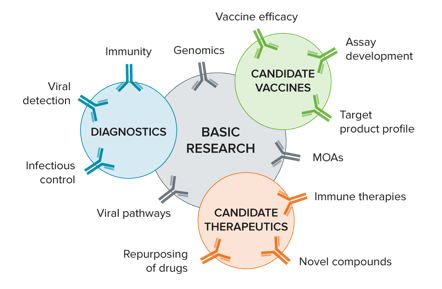
Our customers are developing potential therapies for COVID-19 including:
- Vaccines
- Recombinant proteins, including monoclonal antibodies
- Repurposing of drug molecules already approved, or in clinical trials, for other viruses
- Developing diagnostic and research tools using antibodies
Applications related to COVID-19 and critical research
Learn more about how our technology can help support your research of COVID-19 cellular responses and vaccine development. We’ll address common applications used in infectious disease research including ELISAs and cell line development to Viral Pathways and offer resources to aid in your research.
Coronavirus vaccine research:
- Cell Count/Cell Viability
- Cell Line Development
- Protein/Antigen Binding Affinity
- Protein/Nucleic Acid Quantification (Non-specific)
- Protein/Nucleic Acid Quantification (Specific)
- Viral Neutralization
- Viral Pathogenesis (Viral Pathways)
- Viral Titer

Vaccine development workflow
Vaccine development workflows vary depending upon the platform (e.g. inactivated virus vs. DNA vaccine) chosen, each having its own advantages. In order to increase the likelihood of success against the infectious agent, CEPI, the Coalition for Epidemic Preparedness Innovations, and many other organizations promote diverse approaches during a pandemic.
We’ve created a variety of virus-related workflows – from antigen/immunogen and antibody discovery to stable cell line development. Below is a general workflow for vaccine development using recombinant proteins as the immunogen, referencing the systems to aid in your research.

- Screen for binding to virus-specific immunogen – Screen virus-specific immunogens through binding using phage display
- Immunogen Characterization – Characterize lead immunogen candidates
- Protein (immunogen) Engineering – Optimize lead immunogen sequence and structure to enhance anti-viral function
- Cell Transfection and Cloning – Introduce lead immunogen sequence into cells for protein expression
- Screen for Quality Attributes, including Titer – Screen transfected cells to identify clones with high protein yield and desired quality attributes
Related News & Articles
Imaging system from Molecular Devices helps Recursion produce the largest publicly available set of human cellular morphological data for COVID-19 therapeutic research
Technology Networks Spotlight
The Science Explorer interviews Justin Dranschak from Molecular Devices to discuss how we are supporting COVID-19 scientists in their analysis of immunology, candidate vaccines, therapeutics, and diagnostics.