Advancing scientific discovery
Passionate about equipping researchers with next-generation technology, inspiring innovative solutions that advance scientific discovery, to improve the quality of human life worldwide.
A life science company
Four decades dedicated to enabling life science labs of the future, where innovative technology and novel research converge.

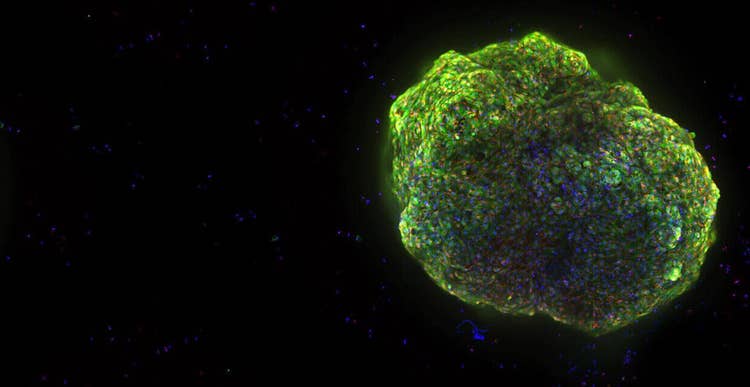
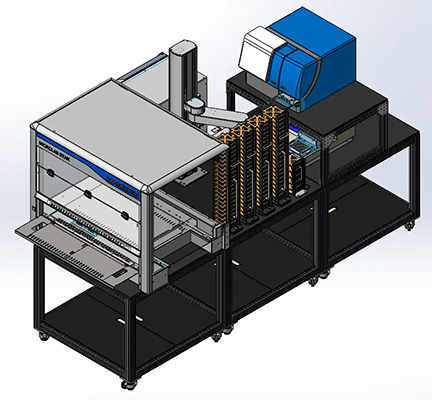
State-of-the-art automated 3D imaging workcell
The Organoid Innovation Center at Molecular Devices combines cutting-edge technologies with novel research methods to address key challenges of scaling complex 3D biological models.
The collaborative space brings customers and researchers into the lab to test automated workflows for organoid culturing and screening, with guidance from in-house scientists.
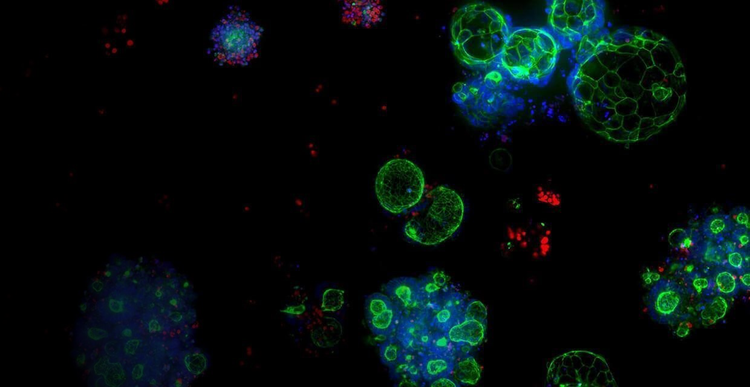
3D Biology
Pioneering a new future of drug discovery rooted in 3D biology will empower researchers to personalize therapies, reduce a medicine’s time to market, and enhance quality of life for patients around the world.
Leveraging proprietary bioprocess technology to produce reliable and predictive organoids at scale to innovating the cell culture system of the future, we believe in the revolutionary promise of 3D biology.

Cell Engineering
The addition of our colony picker platform expands the Center’s state-of-the-art methods and technologies to include automated workflows for synthetic biology, antibody discovery, and cell line development.
And, with the recent launch of CloneSelect® Imager FL with multi-channel fluorescence for day zero monoclonality verification and confirmation of CRISPR edits, provides a wide range of cell and gene therapy applications.
Lab Automation & Customization

From our PhD-level technical support staff to our expert-trained and certified field service engineers, we guide scientists through customized automation solutions that are uniquely designed to meet the specific needs of their assay, method, or protocol.
Explore our integrated end-to-end, high-throughput screening solutions:
We empower scientists to advance discovery
Automated, end-to-end solutions span research disciplines to help scientists discover, scale, and industrialize biology.
SpectraMax® Microplate Readers and SoftMax® Pro Software
For 40 years, we have partnered with scientists to help them achieve landmark discoveries. Our microplate readers and software are the industry’s most cited and have empowered life science researchers to advance protein and cell biology.

Streamline your GxP compliance journey for GMP/GLP labs
A leader in comprehensive GxP compliance solutions, we combine our SpectraMax® microplate readers, SoftMax® Pro GxP software, SpectraTest® Validation Plates and expert IQ/OQ/PM services to help laboratories operating under GMP and GLP regulations follow FDA or regional regulatory guidelines with confidence.
Save on a bundle: Microplate reader and washer
Improve your lab’s efficiency, get more data, and decrease time to discovery with an automated microplate reader and washer solution:
Lab automation for high-throughput plate-based assays
Explore our fully-integrated, automated workflow solutions for cell and biochemical assays. Our scalable ELISA Workcells provide walkaway time, increasing throughput, effectiveness and efficiency of the assay procedure, and reproducibility.


Introducing the revolutionary CellXpress.ai™ Automated Cell Culture System
An Al-driven cell culture innovation hub, the CellXpress.ai system automates processes, improves workflows, and makes assays more reliable and reproducible with machine learning-assisted monitoring, feeding, imaging and scheduling.
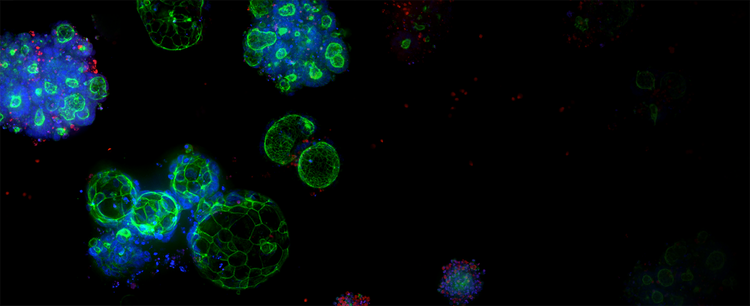
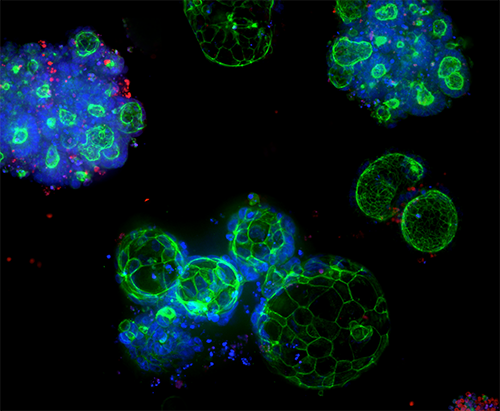
3D Ready™ Organoid Expansion Service
Quality-controlled organoids are manufactured at scale for high-throughput screening, leveraging proprietary bioreactor and bioprocess technology to produce reliable and predictive patient-derived organoids (PDOs).


Automated, turnkey platform with BioAssemblyBot® 400 bioprinting technology
Out-of-the-box integration of the enhanced BAB400 with the ImageXpress® Confocal HT.ai High-Content Imaging System and IN Carta® Image Analysis Software for an optimized end-to-end workflow solution for your 3D biology research.


ImageXpress® High-Content Confocal Imaging System with AI-driven Technology
Ranging from automated digital microscopy to high-throughput confocal imaging systems with water immersion objectives, proprietary spinning disk technology, 7-channel laser light source, and machine-learning software analysis workflow.

Clone screening systems for colony picking and single-cell isolation
The QPix® 400 Microbial Colony Picker, ClonePix® 2 Mammalian Colony Picker, and CloneSelect® FL Imager increase throughput and consistency across cell line development, monoclonal antibody discovery, and synthetic biology workflows.

Automated solutions for high-throughput clone screening
Fully-integrated, lab automation solution for molecular cloning, antibody discovery, and monoclonality. Our automated clone screening workflows integrate laboratory devices to increase your throughput and efficiency while reducing human interaction.

Single-cell dispensing and screening for monoclonality verification.
Award-winning DispenCell™ Single-Cell Dispenser and the CloneSelect® Imager FL – our bundled solution offers unparalleled precision and efficiency in cell line development and monoclonality verification at day zero.

Plasmid Mini-Prep Starter Bundle
A fully automated platform designed for researchers aiming to streamline their plasmid preparation process. From transformation to plasmid purification, achieve low to medium throughput with validated protocols, all in an entry-level automation setup with the potential to expand.
- QPix® XE Microbial Colony Picker
- SpectraMax® i3x Microplate Reader
- Integrated with liquid handler, shaking incubator, robotic arm, and plate hotel
We’re scientists supporting scientists
We empower scientists to advance discovery, driving earlier diagnoses and safer therapeutics for patients.

3D Biology: The paradigm shift in next-gen drug discovery
In the midst of an evolving drug discovery paradigm, researchers worldwide are transitioning their compound screens away from 2D cell cultures and animal models to more complex, human-relevant 3D systems like organoids. Experience our interactive infographic as it takes you deeper into why the industry is embracing this next generation of drug discovery and the innovations supporting scientists in their 3D biology journey.
Drug Discovery & Development
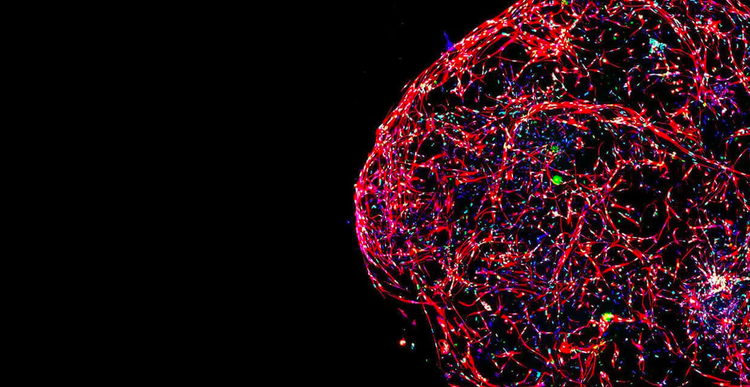
The drug discovery landscape is shifting, with more scientists centering cell line development, disease models, and high-throughput screening methods around physiologically-relevant 3D cell models.
The reason for this is clear: Using cellular model systems in research that closely mimic patient disease states or human organs can bring life-saving therapeutics to market – faster.
3D Cell Models
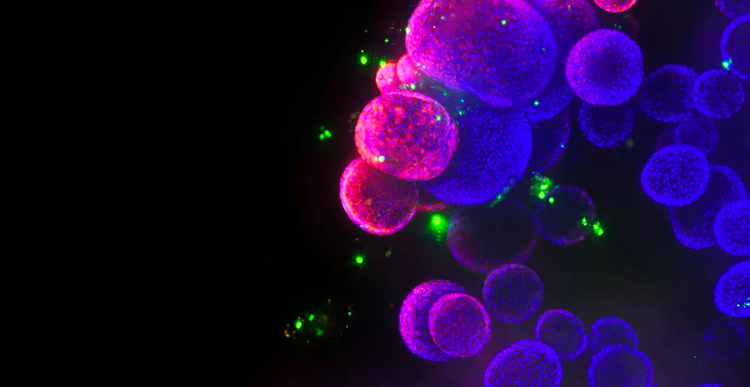
With our automated 3D cell culture and bioimaging screening solutions, we are helping reshape the future of drug discovery. Supported by our technology and organoid development protocols, researchers can now advance and scale screening methods for physiologically-relevant 3D models that more closely mimic patient disease states and human organs, leading to faster drug development and approval.
Cell Line Development
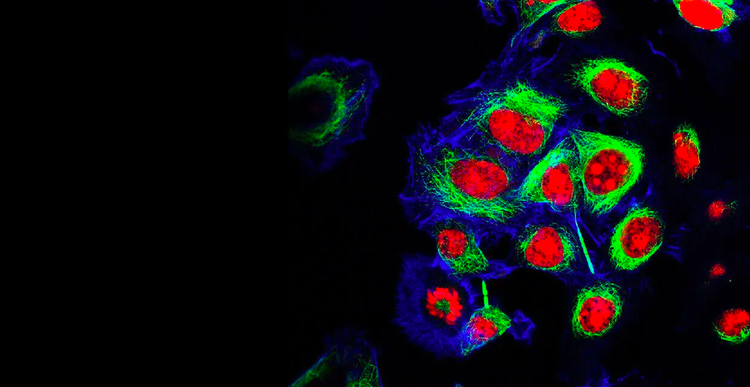
Our cell line development customers accelerate time to market for life saving monoclonal antibodies (mAbs) and genomic medicine, as well as perform groundbreaking research in cell and gene therapies, genetic engineering, personalized and precision medicine, synthetic biology, RNA and DNA-based vaccines, and so much more – made possible by our innovative technology and rich expertise.
Drug Screening
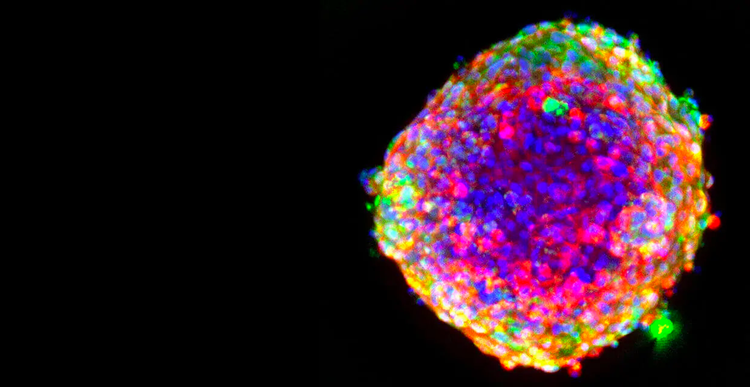
For every drug that makes it to the finish line, another nine don’t succeed. This alarming failure rate can be traced to reliance on 2D cell cultures that don’t closely mimic complex human biology, often leading to inaccurate predictions of a drug’s potential and extended drug development timelines.
A critical step in the drug discovery process, drug screening and toxicity assessment uncover the effects of potentially life-saving compounds. Shifting to cell-based testing allows multiple chemicals to be tested rapidly and better represents human biology.
